Researchers from Northwestern Medicine discovered a new drug that was highly effective in lowering the signs and symptoms of moderate-to-severe eczema. This is the first trial to use a biologic therapy (monoclonal antibody) instead of immune-suppressing drugs to treat moderate-to-severe eczema in infants and children aged 6 months to 5 years. More than half the children experienced at least a 75 per cent reduction in eczema symptoms, highly significant reductions in itching, and improved sleep after a 16-week course of dupilumab, a drug that targets a crucial immunological system in allergens.
This is the first massive, random, placebo-controlled trial of a monoclonal antibody for any skin condition, including eczema, in kids as young as six months. The study, which covered 31 locations across Europe and North America, will be released in The Lancet on September 15. According to the lead study author Dr Amy Paller, chair of dermatology at Northwestern University Feinberg School of Medicine and an attending physician at Northwestern Memorial Hospital, “preschoolers who are constantly scratching, awake multiple times a night with their parents, irritable and markedly limited in their ability to do what other children their age can do improved to the extent that they sleep through the night, change their personalities, and have a normal life — as babies and children should.”
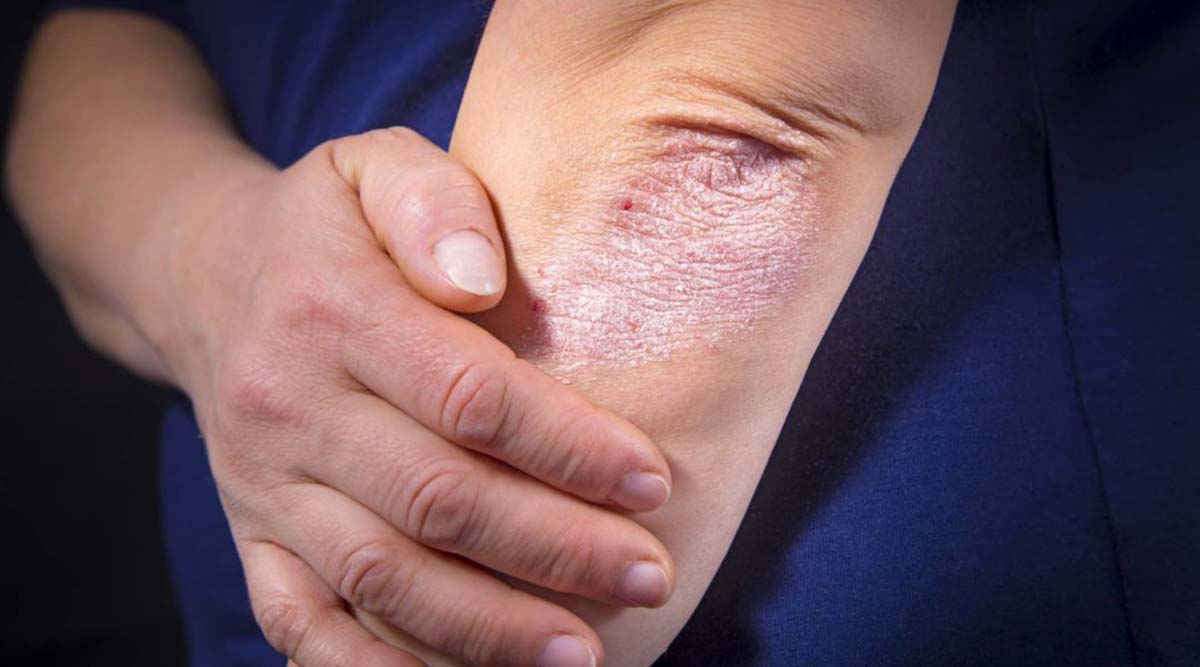
Eczema, also known as atopic dermatitis, is a chronic inflammatory skin condition that causes itching, red, dry skin, and frequent weeping. It can have a significant impact on the life of both the patient and their family. More than one-fifth of all children under the age of six are thought to have eczema and 85 to 90 per cent of those who have the condition as a whole experience its commencement in the first five years of life.
The crippling itch of the kids causes sleep disruption, poor neurocognitive growth, and, on average, a full night of lost sleep every week. Infants and young children who suffer greatly from this disease will live far better lives if they can take this medication, according to Paller. “There is a lot more to atopic dermatitis or eczema than just scratchy skin. It is a fatal condition. Severe eczema has a quality of life that rivals many illnesses that are potentially fatal, not just for the child who has it but also for the parents.”
This study led to the availability of this drug for infants and preschoolers as young as 6 months. According to Paller, it has “an amazing safety profile” and doesn’t even need any laboratory tests before the prescription is started. The remaining one-third or more of young children with eczema have moderate-to-severe illness and require more active therapy, even if half to two-thirds of them have mild symptoms that can be managed with steroid ointment and moisturisers.
“Up to now, all we have had to treat more severe eczema is immune-suppressing medications, such as oral steroids, which we try to avoid in children because they are associated with so many side effects and thus are not a preferred treatment for a chronic skin disease,” Paller said. “The potential long-term impact on the development of the immune system in young children is also of concern with these immunosuppressants.” The first “biologic” treatment to treat eczema in a targeted manner or a focused attack on only what researchers have discovered is causing the disease’s skin symptoms, has recently been accessible and is called dupilumab. This medication was determined to be efficient and secure in tests involving adults, adolescents, and other school-aged children.
However, Paller noted that children under 5 were not assessed for safety and could not receive this drug. The child receives a monthly shot of the drug from a parent or medical professional.
“The effect for most of these younger children is dramatic and at least as good as we’ve seen with the risky immunosuppressant medications,” Paller said. Potential added benefit by treating associated allergies
Although it has been proven to be helpful in treating asthma, gastrointestinal allergy symptoms, and other allergy-related issues, this medicine is not currently licenced for use in newborns and young children. In fact, more than 80% of children in this experiment had already developed at least one allergic disease, such as asthma or a food allergy, by the time the dupilumab was started. 66% of children in this trial had eczema during the first six months of life.
“By treating more aggressively to calm the immune system activation in these young children with early, severe eczema, we may also reduce the risk of their developing a range of allergic problems, changing their life beyond improving eczema,” Paller said. “These associated allergic issues most often begin after eczema starts.” Every four weeks for 16 weeks, children were randomly assigned to receive either a placebo injection or the weight-based dose of dupilumab. The only kids who could enrol were those whose topical treatments weren’t working well on them, and even then, their severity level had to be high. Paller added that as a consequence of the study, researchers and medical professionals can begin to comprehend the connections between eczema and a number of allergy problems and can think about whether to use this medicine for additional conditions that affect these very young children.

